Medigen Ankle Arthroscopy Drape for Minimally Invasive Ankle Surgery
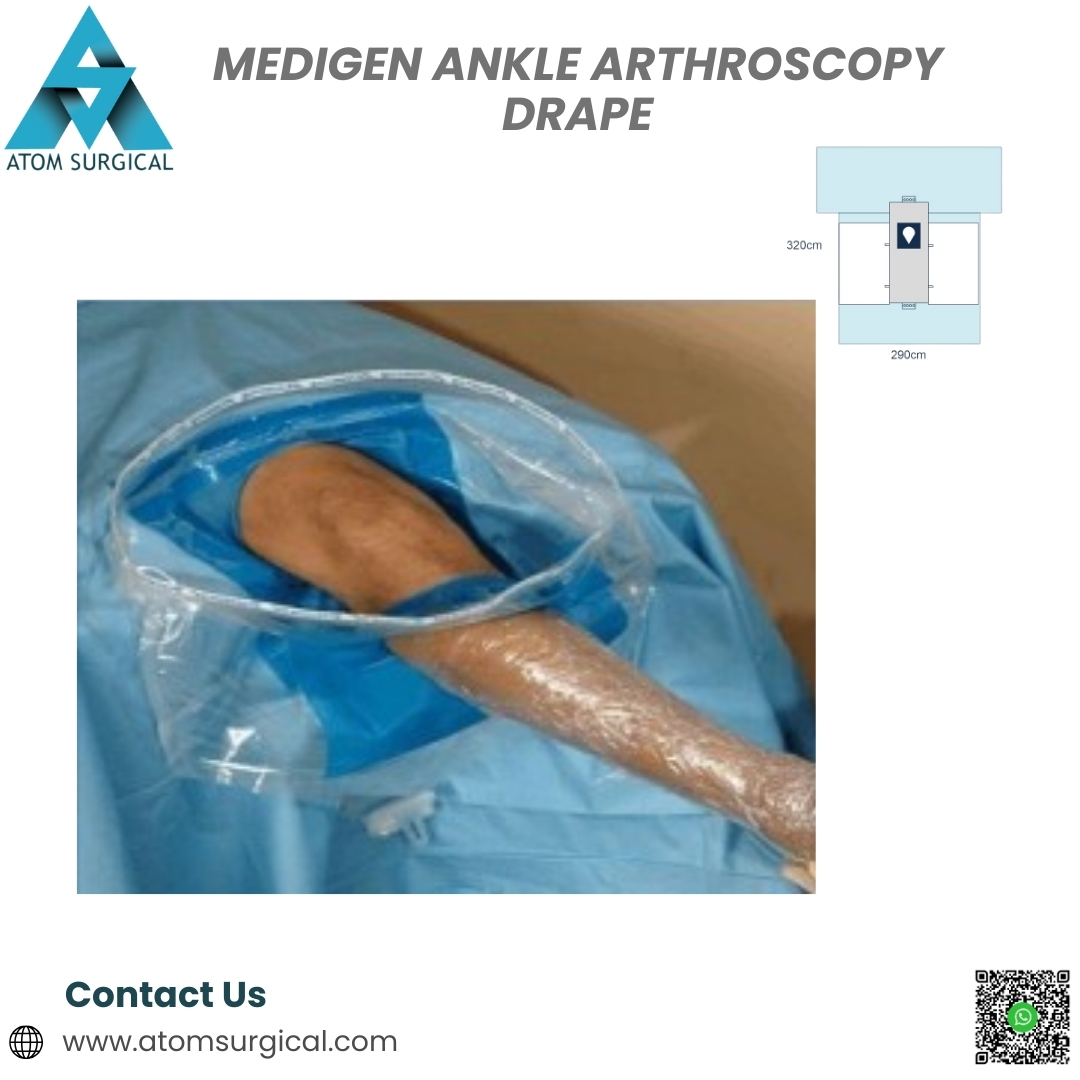
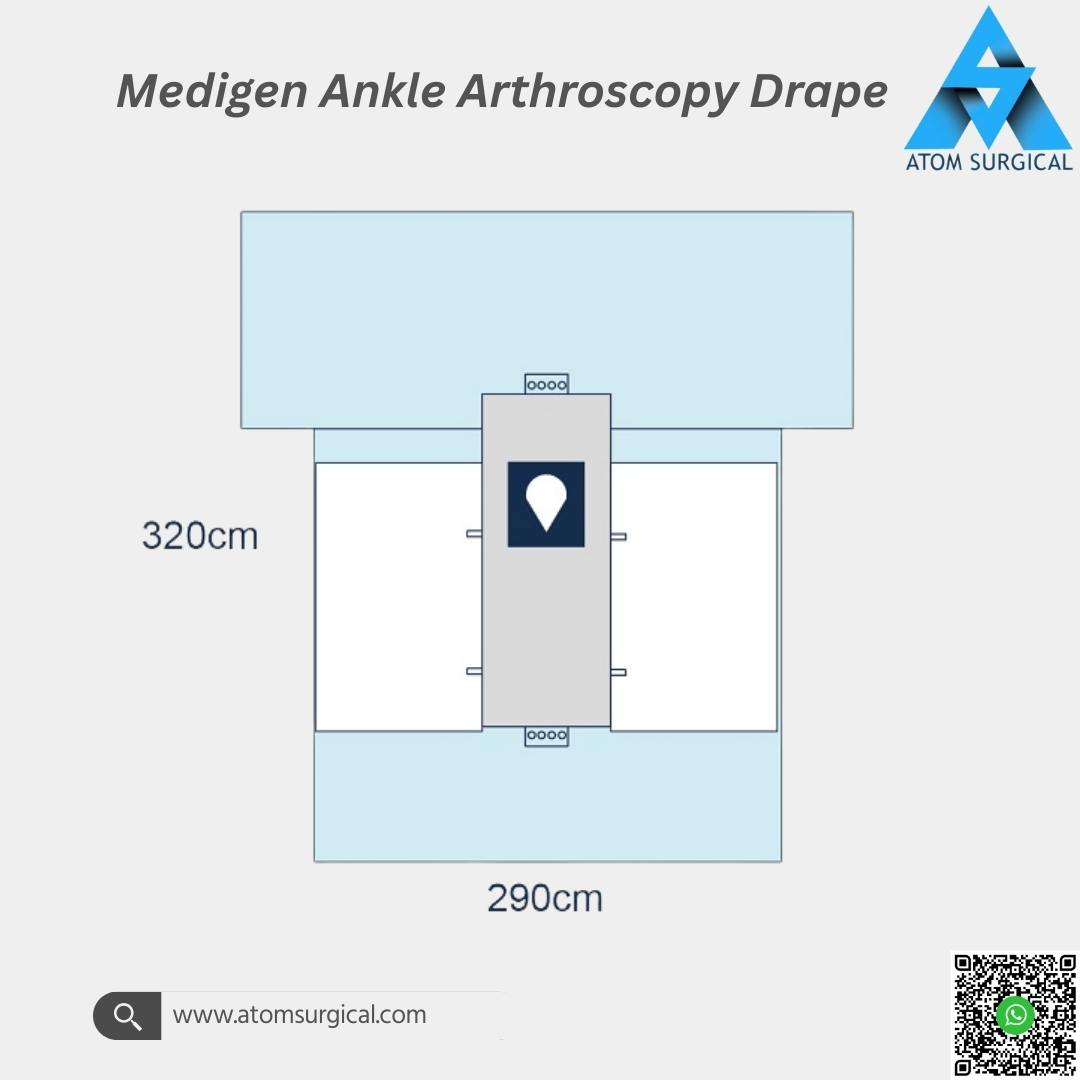
The Medigen Ankle Arthroscopy Drape is a single-use, EO-sterilized orthopaedic drape engineered for diagnostic and therapeutic ankle arthroscopy procedures.
It supports surgeons by maintaining a clean, dry, and sterile operative field, even during procedures involving continuous saline irrigation.
Advanced Fluid Management & Barrier Protection
Ankle arthroscopy requires precise control of irrigation fluids in a compact surgical area.
This drape is manufactured using premium SMS and laminated non-woven materials and offers:
-
High fluid and blood resistance
-
Integrated fluid collection pouch (optional)
-
Reliable microbial barrier protection
-
Low lint performance for clear arthroscopic imaging
Recommended Surgical Applications
The Medigen Ankle Arthroscopy Drape is suitable for:
-
Diagnostic ankle arthroscopy
-
Ankle impingement procedures
-
Ligament repair and reconstruction
-
Osteochondral lesion treatment (OLT)
-
Sports injury ankle surgeries
Export-Ready Manufacturing & Customisation
Manufactured by Atom Surgical, this drape is:
-
OEM & private-label ready
-
Customisable in size, fenestration, and pouch configuration
-
Packed for international export logistics
-
Supported with tender and regulatory documentation