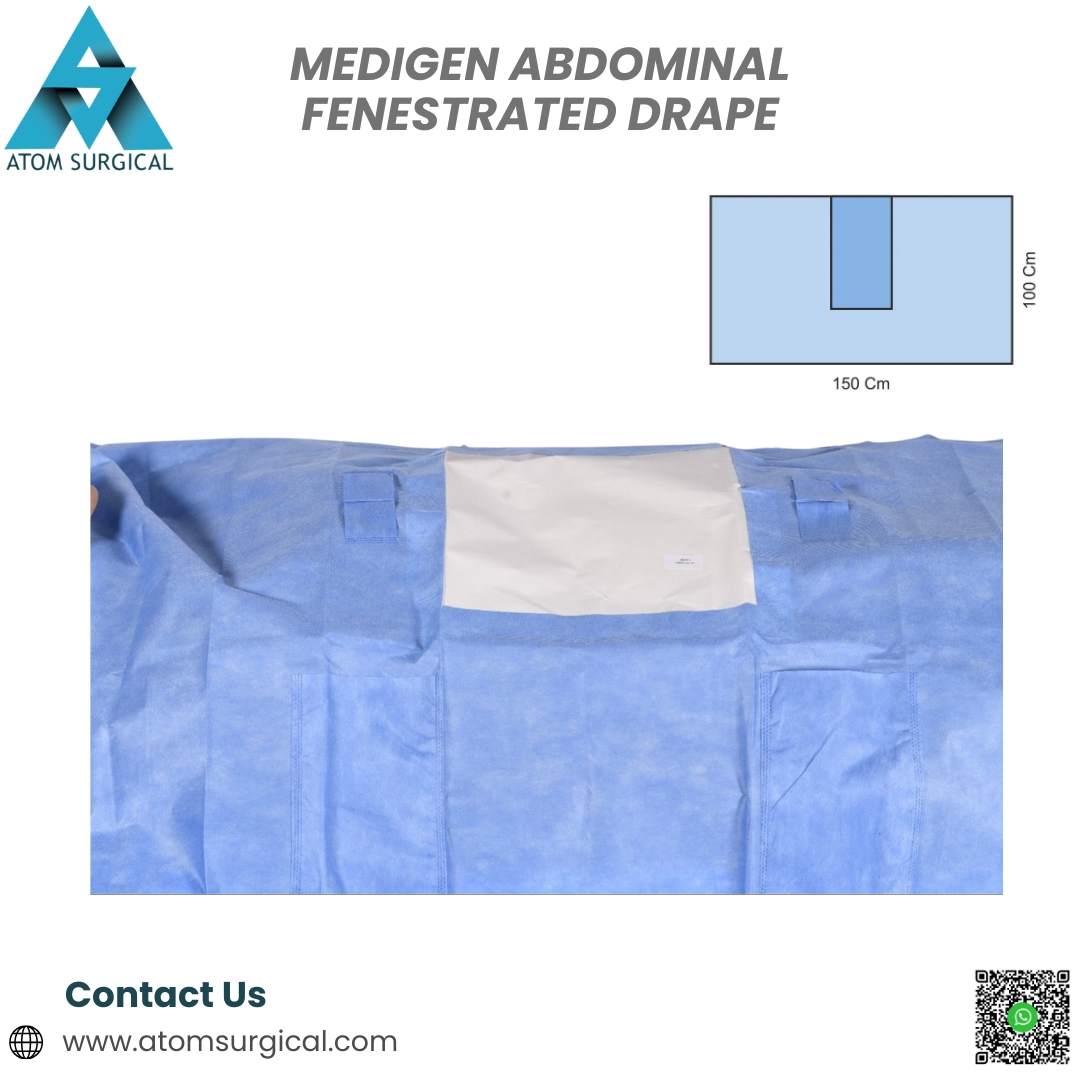
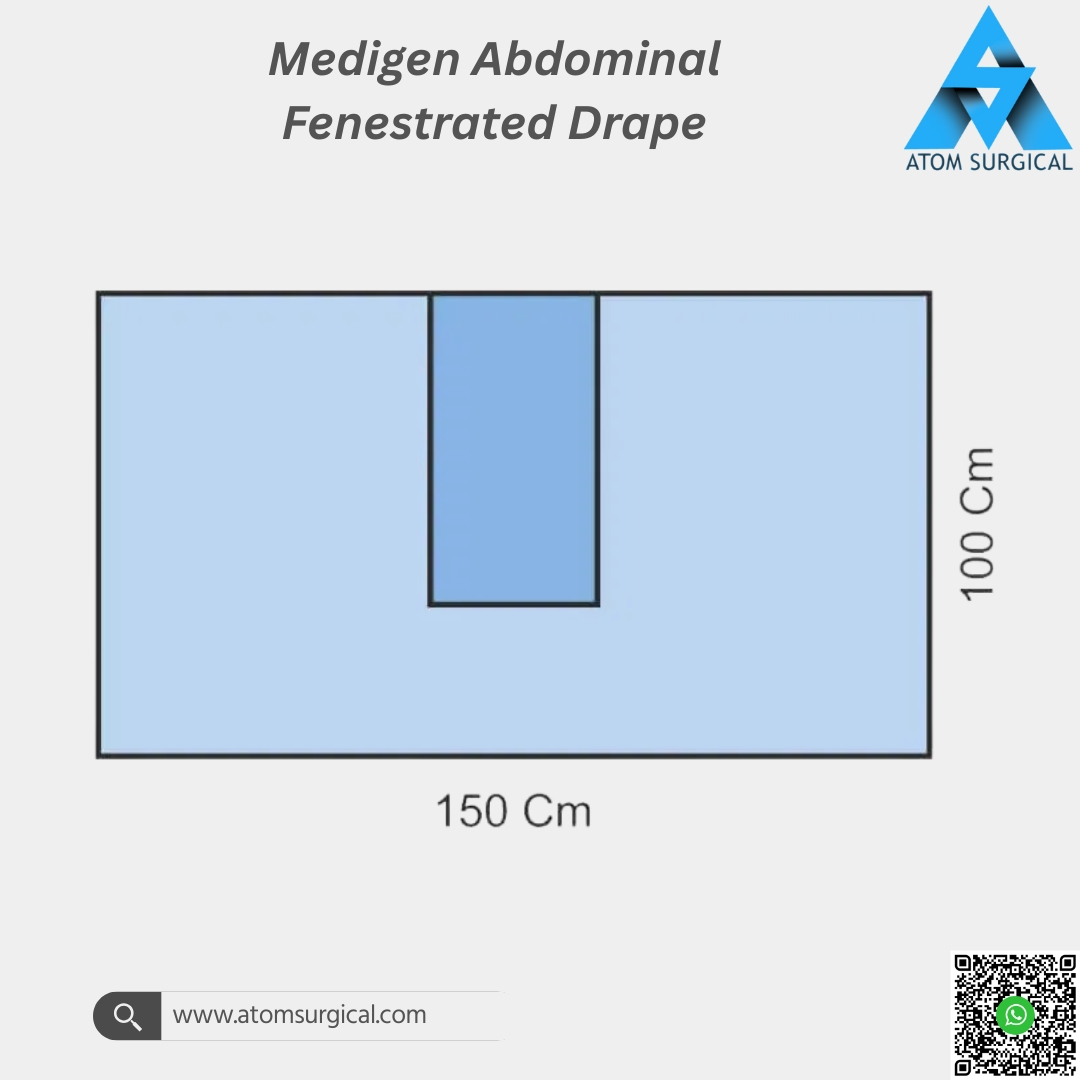
Medigen Abdominal Fenestrated Drape for Open Abdominal Surgery
The Medigen Abdominal Fenestrated Drape is a single-use, EO-sterilized surgical drape developed for abdominal surgical procedures that require precise operative access and strict aseptic control.
Manufactured under controlled quality systems, this drape ensures consistent performance, reduced contamination risk, and improved surgical workflow.
High Barrier Protection with Precision Fenestration
Abdominal surgeries often involve:
-
Large operative fields
-
High blood and fluid exposure
-
Long surgical duration
This fenestrated abdominal drape is manufactured using premium SMS and laminated non-woven materials, offering:
-
High fluid and blood resistance
-
Reliable microbial barrier protection
-
Low lint performance for a clean operative field
-
Precisely cut fenestration for accurate incision access
Recommended Surgical Applications
The Medigen Abdominal Fenestrated Drape is suitable for:
-
General abdominal surgeries
-
Laparotomy procedures
-
Gastrointestinal surgeries
-
Colorectal and hernia surgeries
-
Emergency and elective abdominal operations
Export-Ready Manufacturing & Customisation
Manufactured by Atom Surgical, this drape is:
-
OEM & private-label ready
-
Customisable in size and fenestration shape
-
Packed for international export logistics
-
Supported with tender and regulatory documentation