Medigen Uterine Drape for Safe & Hygienic Gynecological Procedures
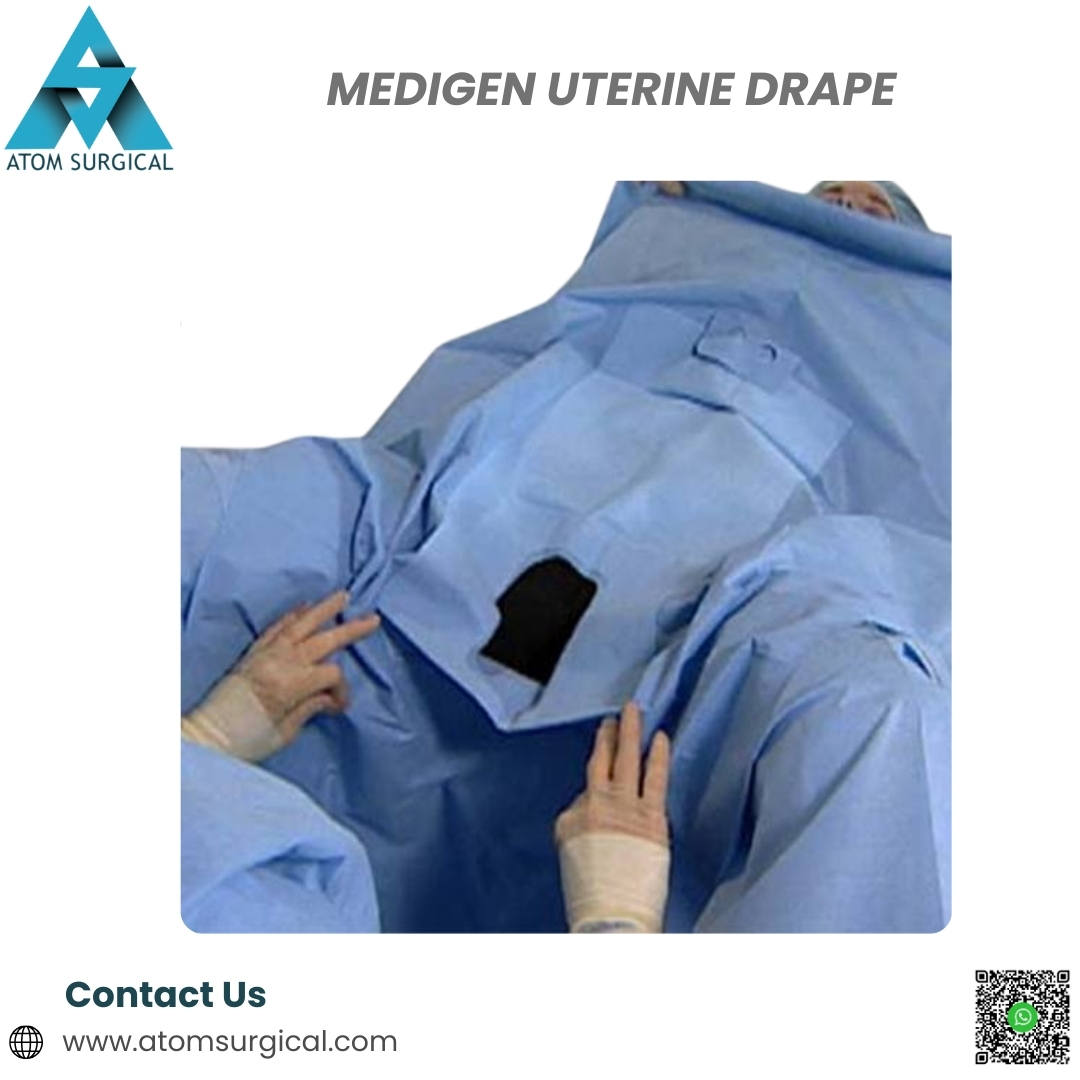
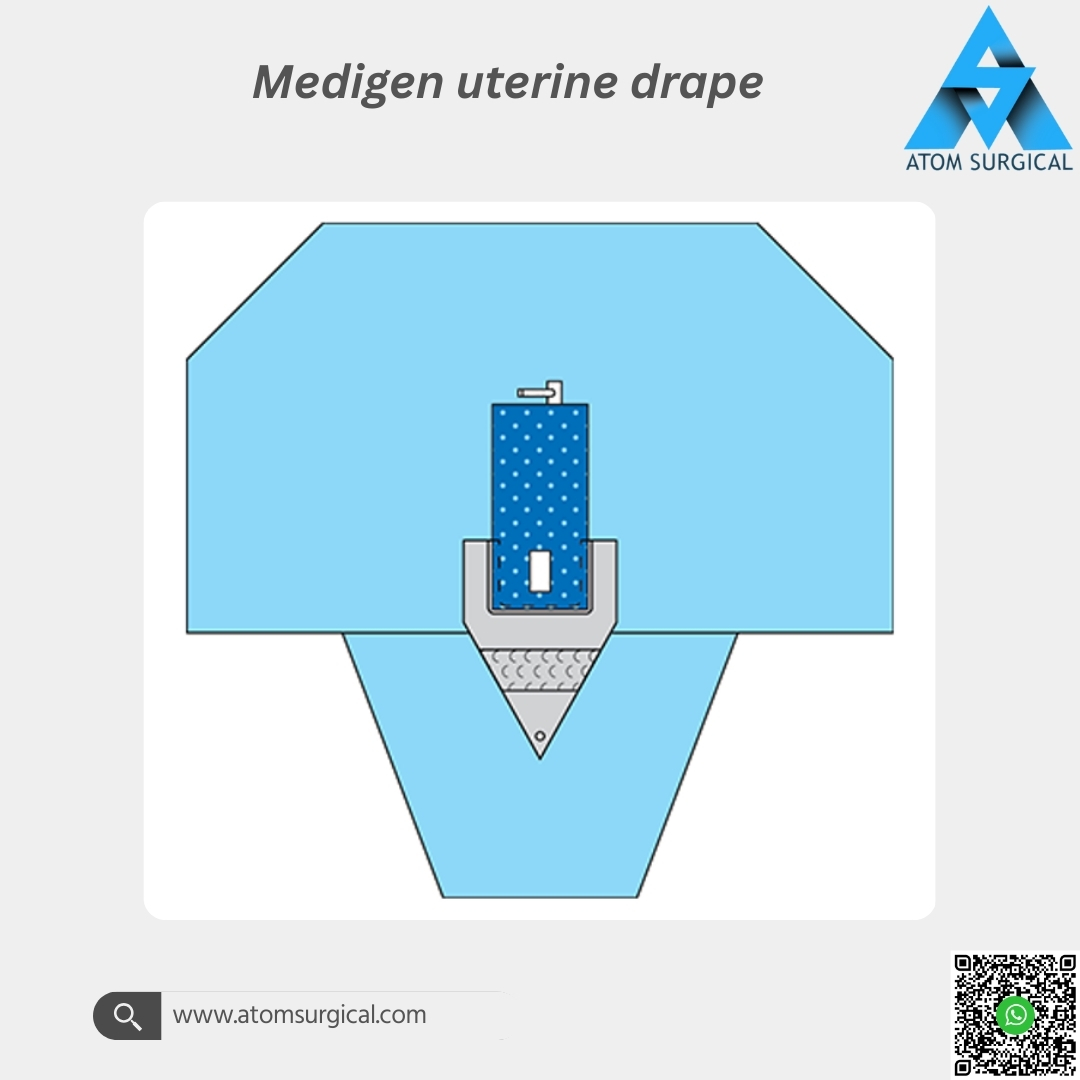
The Medigen Uterine Drape is a single-use, EO-sterilized gynecology surgical drape engineered to provide effective isolation of the uterine and surrounding operative area during diagnostic and operative gynecological procedures.
Manufactured under strict quality management systems, this drape ensures reliable barrier protection, ease of use, and reduced contamination risk in high-turnover clinical environments.
High Barrier Protection & Fluid Control
Uterine procedures often involve:
-
Exposure to blood and body fluids
-
Close proximity to sensitive anatomical areas
-
Strict infection-control requirements
This uterine drape is manufactured using premium SMS and laminated non-woven materials, offering:
-
High fluid and blood resistance
-
Reliable microbial barrier protection
-
Low lint performance for a clean operative field
-
Comfortable patient-contact surface
Recommended Clinical Applications
The Medigen Uterine Drape is suitable for:
-
Uterine surgeries and procedures
-
D&C (Dilatation & Curettage)
-
Hysteroscopy support procedures
-
Gynecological examinations and minor surgeries
-
OB-GYN clinics and hospital gynecology OTs
Export-Ready Manufacturing & Customisation
Manufactured by Atom Surgical, this drape is:
-
OEM & private-label ready
-
Customisable in size, fenestration, and absorbent area
-
Packed for international export logistics
-
Supported with tender and regulatory documentation