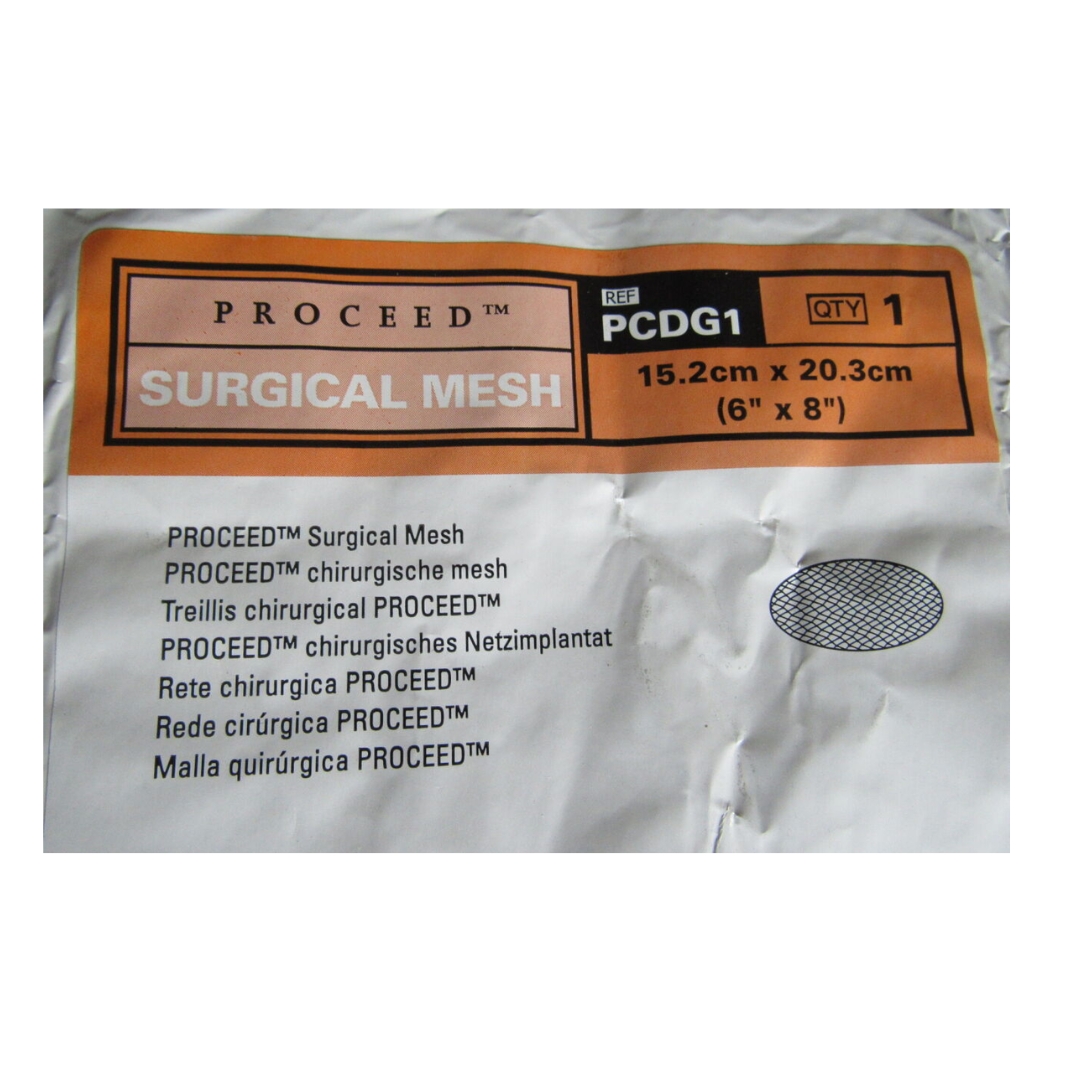
PROCEED Surgical Mesh is a sterile, thin, flexible, mesh implant. It is comprised of an absorbable oxidised regenerated cellulose (ORC) fabric, and a non-absorbable polypropylene mesh that is surrounded by an absorbable polydioxanone polymer component. After absorption of the absorbable components, the polypropylene mesh remains. PROCEED Surgical Mesh is a long term implantable device. This device is MR Safe.
PROCEED Surgical Mesh is indicated for the repair of abdominal wall hernias and abdominal wall deficiencies that require the addition of a reinforcing material to obtain the desired surgical result. Suitability of this device is determined by the surgeon and the selection of mesh for any given patient is a function of numerous factors including, but not limited to, the patient’s past medical and surgical history, current medical condition (i.e., comorbidities), surgical technique, and size and location of the hernia.